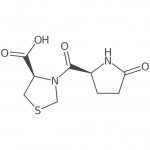
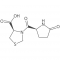
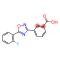
Emeishan Hongsen Pharmaceutical Co., LTD.is a national high-tech enterprise specializing in the fields of peptide fragments,protected amino acids,pharmaceuticalintermediates,active pharmaceutical ingredients(APls), full life-cycle Our capabilities include:CMO/CDMO services,a GMpqualitysystem (audited by domestic and international clients),an in-house R&D Technology Center(Provincial Enterprise Technology Center),the Leshan SyntheticBiology Technology Engineerng Research Center,and one stop services covering process optimization,R&D,manufacturing,ana sales.Founded on Feb18,2011,we have 150+ employees and 70+ reactors(100L-6300L) Flexible collaboration models are available.Our company also has passed the U.S. FEI certification (FEI NO.3017125134), Deng & Bradstreet Certification (D-U-N-S NO.: 417773775), ISO 9001:2015 Quality Management System Certification, ISO 45001:2018 Occupational Health and Safety Management System Certification, ISO 14001:2015 Environmental Management System Certification, implementation of GMP management. At the same time, our products have passed the GMP audit and evaluation of Sichuan Pharmaceutical Industry Association, with more than 26 invention patents.